close
Choose Your Site
Global
Social Media
Product Advantage
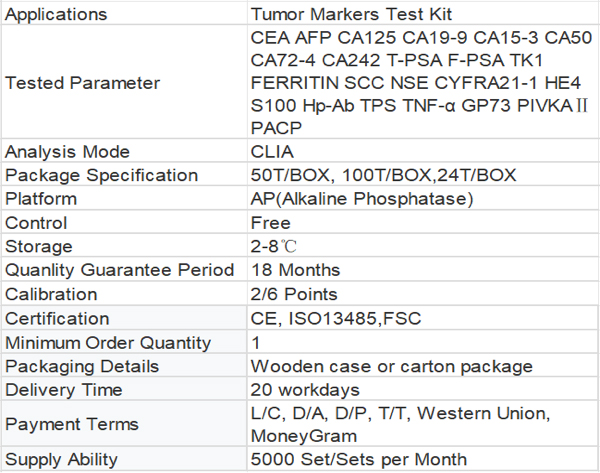
1Extensive testing program: CEA AFP CA125 CA19-9 CA15-3 CA50 CA72-4 CA242 T-PSA F-PSA TK1 FERRITIN SCC NSE CYFRA21-1 HE4 S100 Hp-Ab TPS TNF-α GP73 PIVKAⅡ PACP
2Simple Physical Examination Package:Three tumor screenings:CEA AFP CA199; Male Tumor Five Test:AFPCEACA199TPSAFPSA;Female Tumor Five Test: AFPCEACA199CA125CA153;
3For middle-aged and older people over 40 years old:male: AFPCEACA199CA50TPSAFPSANSECYFRA21-1; female: AFPCEACA199CA50CA125CA153NSECYFRA21-1
4Suitable for people in the age group of 30-50 years old who have a family history of cancer, smoke, drink alcohol, live an irregular life with a lot of stress, and have a bad night's sleep:male: AFPCEACA199CA50SCCCA724CA242TPSAFPSAFERRNSECYFRA211; female: AFPCEACA199CA50SCCCA724CA242CA125CA153FERRB-HCGHE4NSECYFRA211 5Lung Cancer Package:CEANSECYFRA21-1ProGRPSCC
Technical Parameters
Product Uses
Tumor screening physical examination is necessary to allow early detection and early treatment of the disease, and the cure rate of early-stage cancer is much higher than that of middle and late-stage cancer.
Recommended Screening Crowd
Age:After 30 years old, such as testicular cancer, prostate cancer, stomach cancer, colorectal cancer, breast cancer, ovarian cancer, the screening of these tumors should be taken seriously.
Family history of tumor.
People with long-term smoking history.
Work is more tiring, high pressure, often stay up late, irregular diet, or the work process needs to be frequently exposed to carcinogens need to be regular tumor screening physical examination according to occupational characteristics.
People who are easy to get angry, angry, and depressed for a long time will easily lead to the nerve, endocrine and immune system being in a state of high hyperactivity and tension for a long time, which will easily induce cancer.
Product Operate GuideProduct Operate Guide
1. Alpha-fetoprotein
Alpha-fetoprotein is a glycoprotein. Normally, it comes from embryonic stem cells and disappears two weeks after the fetus is born. If the patient's liver cells become cancerous, with the development of the disease, the alpha-fetoprotein in human serum will increase sharply. , which is a specific clinical indicator for the diagnosis of primary liver cancer.
2. Carcinoembryonic antigen
Elevated carcinoembryonic antigen may be due to colorectal cancer, pancreatic cancer, gastric cancer, breast cancer and other cancer diseases, but smoking, pregnancy, diabetes and other diseases may also lead to elevated carcinoembryonic antigen in patients, which is not specific to malignant tumors Sexual markers, but can be used to aid in diagnosis
3. Carbohydrate antigens 199
Elevated carbohydrate antigen 199 is more common in pancreatic cancer, which plays an important role in the differential diagnosis and condition monitoring of pancreatic cancer. It can also be seen in gallbladder cancer, liver cancer, colon cancer and other diseases.
4. Cancer antigen 125
Cancer antigen 125 is mainly used to assist in the diagnosis of malignant serous ovarian cancer and epithelial ovarian cancer, and can also be found in the diagnosis of breast cancer, pancreatic cancer, gastric cancer and other diseases.
5. Tumor antigens 153
Elevated tumor antigen 153 is more common in breast cancer, ovarian cancer, pancreatic cancer, liver cancer and other diseases.